Spotlight on
EMGE
EGME or 2-Methoxyethanol (CAS 109-86-4) belongs to the glycol ether group of solvents. It is a clear, colourless liquid with an ether-like odour. The primary use is as intermediate or process chemicals under controlled conditions in the manufacturing of active pharmaceutical ingredients (APIs).
What’s new
EGME will not be included in annex XIV of REACH, meaning it will not require authorisation for use in the EU.
In autumn 2021, the European Commission proposed not to include EGME in annex XIV of REACH. The REACH Committee (Member States) have endorsed this proposal via written vote. On 10th February 2022, the period for the co-legislator ended with no opposition from the European Parliament or the Council, leading to EGME not being included in annex XIV as the outcome.
Following Council decisions, EGME is also one of the 12 substances moved from the Chemical Agent Directive (CAD) in order to include reprotoxic substances in the Carcinogenic & Mutagenic Directive (CMD).
Background
EGME is one of the 8 Glycol Ethers classified for reproductive toxicity 1B under the CLP Regulation. The so-called “OSPA Charter”, a voluntary agreement, was set up in 1996 and forbids, under penalty of non-delivery, all uses of glycol ethers classified as reprotoxic in any product sold to the public and strictly limits the use to industrial applications, for which no substitute has been found so far. All customers must ensure that Occupational Exposures/Emissions are within the legal constraints. The charter must be signed by buyers (from direct customers) and any distributor involved. The producers oblige all buyers to reconfirm the application of the charter annually.
- In 2009, an indicative occupational exposure limit value was set for EGME.
- In 2010, EGME was identified as Substance of Very High Concern under Reach and added to the Candidate List
- In 2019, EGME was included in the 9th Recommendation for annex XIV inclusion. Ten years have passed since the Candidate List inclusion and the OEL setting.
Most of the remaining usages today are as intermediates, which would have been out of the scope of authorisation. EGME is an essential solvent in the production of APIs (active pharmaceutical ingredient) at only two companies. Here, EGME is used in compliance with the “OSPA charter”, meaning any exposures from use will be within regulatory occupational exposure standards (0,1 ppm; time-weighted average exposure limit 8h) and the same stringently controlled use conditions for use as a process chemical are expected as for use as an intermediate.
- Downstream User 1: The API is used by a company to produce a life-saving antifungal drug (medicinal product). It is an approved broad-spectrum triazole antifungal agent for the treatment of systemic fungal infections & recognised as an “orphan medicinal product”. The United States Food and Drug Administration, FDA, gave a “Fast track approval” because of its importance.
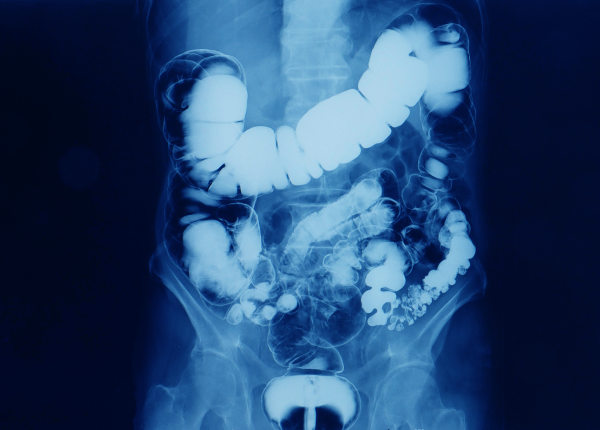
- Downstream User 2: Two APIs are produced for X-ray contrast media, with a global market share of 35-40%, meaning that almost 240.000 patients use those products daily to receive a diagnosis.
The residual level of 2-methoxyethanol in the API is <0.005%. This is in accordance with ICH Q3C (Guideline for residual solvents in pharmaceuticals), which defines maximum limits for solvent residues in active pharmaceutical ingredients (including for 2-methoxyethanol).
Interesting to note: If the API was used to produce the final medication or medical product, the API production as a pre-step would fall under REACH Article 2(5), which exempts from registration, downstream user requirements, evaluation and authorisation substances “used in” medicinal products for human or veterinary use within the scope of Regulation 726/2004, Directive 2001/82 and Directive 2001/83.